India’s First Indigenous & Gender Neutral HPV Vaccine Is Here!
CERVAVAC®
Quadrivalent Human Papillomavirus (HPV Types 6, 11, 16, & 18) Vaccine (Recombinant)

About HPV
The human papillomavirus (HPV) is a common virus that affects both males and females. HPV is highly transmissible, particularly after the onset of sexual activity, and most persons acquire infection at some time in their lives. There are many different types of HPV. Some types can cause health problems, including genital warts and cancers. HPV can cause cervical and other cancers, including cancer of the vulva, vagina, penis, or anus. It can also cause cancer in the back of the throat (called oropharyngeal cancer). HPV 6 and 11, two of the “low-risk” genotypes, cause genital warts, a common benign condition of the external genitalia that causes significant morbidity. HPV 16 and 18, the most common “high-risk” genotypes, cause approximately 70% of all cervical cancers.
CERVAVAC® Indications
● CERVAVAC® helps protect individuals ages 9 to 26 against the following diseases
caused by four types of HPV: cervical, vaginal, and vulvar cancers in females, anal
cancer, and genital warts in both males and females
● CERVAVAC® helps protects against 4 types of HPV: 6, 11, 16, 18.
● Types 16 and 18 are the cause of most cervical cancers globally (more than 70%).
● These types of HPV also cause most anal cancers, and some genital cancers.
● HPV types 6 and 11 cause around 90% of genital warts.
CERVAVAC® is indicated in girls and women 9 through 26 years of age for the
prevention of the following diseases caused by Human Papillomavirus (HPV) types,
included in the vaccine:
• Cervical, vulvar, vaginal, and anal cancer caused by HPV types 16 and 18
• Genital warts (condyloma acuminata) caused by HPV types 6 and 11
And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16,
and 18:
• Cervical intraepithelial neoplasia (CIN) grade 2/3 and Cervical adenocarcinoma in
situ (AIS)
• Cervical intraepithelial neoplasia (CIN) grade 1
• Vulvar intraepithelial neoplasia (VIN) grade 2 and grade 3
• Vaginal intraepithelial neoplasia (VaIN) grade 2 and grade 3
• Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3
CERVAVAC® is indicated in boys and men 9 through 26 years of age for the
prevention of the following diseases caused by HPV types included in the vaccine:
• Anal cancer caused by HPV types 16 and 18
• Genital warts (condyloma acuminata) caused by HPV types 6 and 11
And the following precancerous or dysplastic lesions caused by HPV types 6, 11, 16, and 18:
• Anal intraepithelial neoplasia (AIN) grades 1, 2, and 3
CERVAVAC® stands as India’s pioneering HPV vaccine, cultivated domestically to prevent cervical cancer. This quadrivalent vaccine combats four distinct HPV variants (HPV 6, 11, 16, and 18).
- Pune’s Serum Institute of India (SII) developed the initial Indian cervical cancer vaccine, also known as the human papillomavirus (HPV) vaccine (qHPV).
- The Drugs Controller General of India (DGCI) granted approval for its use.
- This vaccine resulted from collaborative efforts between SII and the Department of Biotechnology (DBT).
Vaccine Composition Information
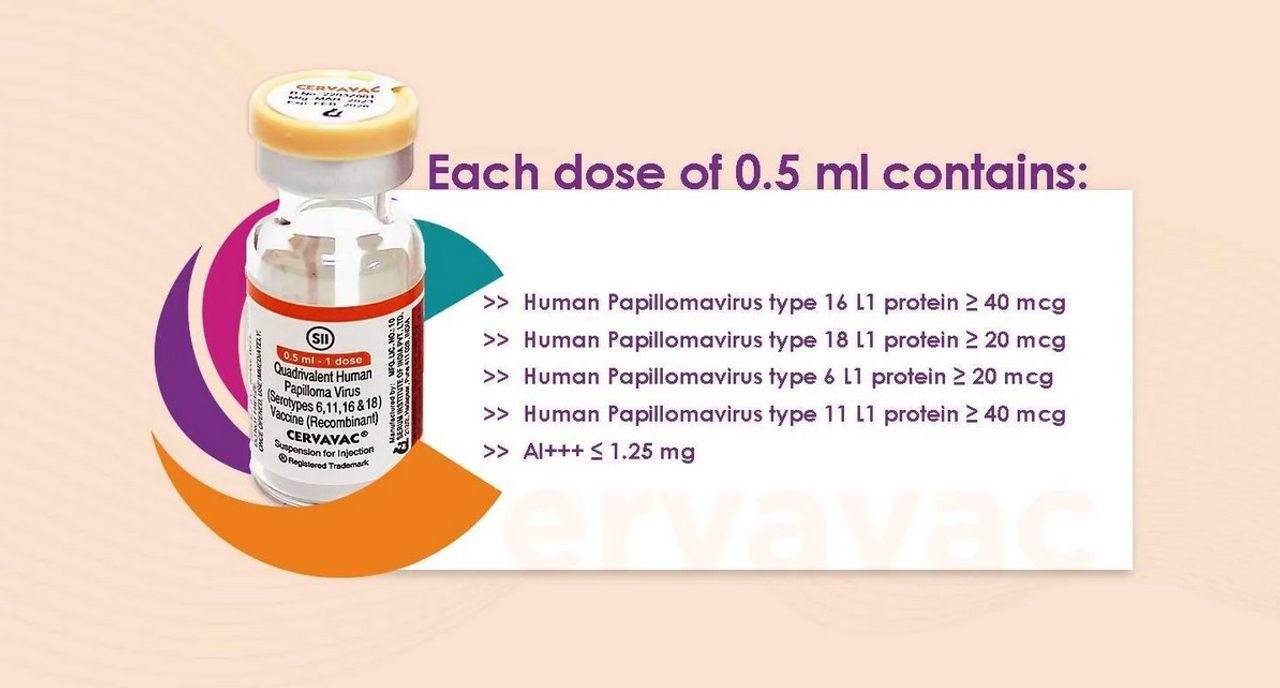
Dosage Info

For vaccine recipients between 9 and 14 years old, CERVAVAC® should be administered according to a 2-dose schedule (0.5 ml at 0 and 6 months).

For vaccine recipients between 15 and 26 years old, CERVAVAC® should be administered according to a 3-dose schedule (0.5 ml at 0, 2, and 6 months).

The second dose should be administered at least one month after the first dose and the third dose should be administered at least 3 months after the second dose. All three doses should be given within a 1-year period.
Copyright © 2023. Serum Institute of India Pvt. Ltd.



